Most Important Considerations
Encouraging tobacco use cessation is one of the few interventions that can alter the natural history of COPD.
Quit rates as high as 25% can be achieved with the investment of sufficient time and resources (e.g., pharmacologic therapy plus clinician-delivered counseling). Clinicians should strive for complete delivery of the “5-As”:1
- Ask every patient about tobacco use at every encounter
- Strongly Advise users to quit
- Assess the patient’s desire and readiness to make a quit attempt
- Assist the patient in their quit attempt
- Arrange in-person or phone follow-up.
Pharmacologic therapy (e.g., nicotine replacement, antidepressants, varenicline) can increase quit rates; however, it is even more effective when combined with counseling interventions. Even counseling sessions as brief as a 3-minute intervention urging a person to quit can measurably increase quit rates. Beyond that, more intense counseling, which may involve more sessions, longer sessions, or both, can increase the effectiveness of counseling interventions, particularly when clinicians are specifically trained to deliver these sessions and provided feedback on their performance.1
Pulmonary rehabilitation is the most effective intervention for improving symptoms and has significant benefits that persist across different severities of COPD and well beyond the immediate rehabilitation period.
GOLD suggests that pulmonary rehabilitation is appropriate for most COPD patients. It is the most effective intervention for improving symptoms, having been shown to improve exercise capacity and quality of life across different severities of COPD [LOE: A]. It is also highly cost effective, with an estimated cost of $2,500-$10,000 per quality-adjusted life year. Patients in GOLD groups B, C, or D should be encouraged to participate in a formal rehabilitation program.1
The GOLD Workgroup suggests that pulmonary rehabilitation programs are comprehensive interventions consisting of, but not limited to, exercise training, education, and self-management training to promote behavior change. The scope and intensity should be individualized for each patient. Programs should last at least 6-8 weeks; however, evidence does demonstrate greater benefit with programs lasting ≥ 12 weeks. Cost and availability are often key determinants in the choice of a program. Comparable home- and community-based rehabilitation programs are considered equivalent to hospital-based programs. Increases in exercise capacity may not necessarily translate into increased daily physical activity.1,6
ACP/ACCP/ATS/ERS recommend that pulmonary rehabilitation programs be prescribed for symptomatic patients with an FEV1 < 50% predicted [LOE: Strong rec, Mod evidence] and considered in patients with an FEV1 > 50% who are symptomatic or exercise-limited [LOE: Weak rec, Mod evidence].4 Rehabilitation addresses many non-pulmonary aspects of COPD, including exercise de-conditioning, social isolation, depression, muscle wasting, and weight loss. Benefits, which persist well beyond the immediate rehabilitation period, include improved exercise capacity and quality of life, reduced symptoms and hospitalizations, and improved survival in patients with COPD. Programs should last at least 6 weeks; however, longer programs tend to be more effective.2
The GOLD guidelines recommend assessment of symptoms, FEV1, and exacerbation risk; however, such an approach has not been shown to improve meaningful outcomes.
The goals of COPD assessment are to determine the severity of airflow limitation, the impact on the patient’s health status, and the risk of future events in order to guide therapy. The GOLD guidelines suggest that disease assessment should reflect the complexity of COPD and, consequently, be based on more than airflow limitation. In the past, it was suggested that clinicians use a simple questionnaire such as the Modified British Medical Research Council tool (mMRC) for the assessment of breathlessness. However, because COPD has multiple extra-thoracic effects, the GOLD guidelines suggest using the more comprehensive COPD Assessment Test (CAT).1
The first step in assessment of a patient with COPD is to perform spirometry and categorize the patient’s severity of airflow limitation. Spirometry should be performed after the administration of an adequate dose of short-acting inhaled bronchodilator to minimize variability.1
Table 1. Spirometric Classification of Airflow Limitation
| Category | Airflow limitation | FEV1 range |
| GOLD 1 | Mild | FEV1 ≥ 80% predicted |
| GOLD 2 | Moderate | 50% ≤ FEV1 < 80% predicted |
| GOLD 3 | Severe | 30% ≤ FEV1 < 50% predicted |
| GOLD 4 | Very severe | FEV1 < 30% predicted |
| Adapted from: GOLD guidelines. URL: http://www.goldcopd.org/. Updated: 2020. Accessed: November 13, 2019. | ||
GOLD advises clinicians to 1) assess the symptoms with the CAT scale (or dyspnea with the mMRC) to determine the patient’s level of symptoms and 2) assess their history of exacerbations, including previous hospitalizations. Based on the severity of symptoms and exacerbation history (which predicts future exacerbation risk), patients can be classified into four groups.
Figure 2. GOLD Assessment
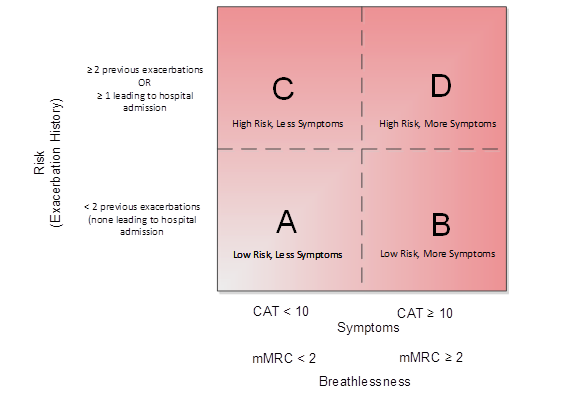
Adapted from: GOLD guidelines. URL: http://www.goldcopd.org/. Updated: 2020. Accessed: November 13, 2019.
Abbreviations: CAT = COPD Assessment Test, mMRC = Modified British Medical Research Council tool
The 2017 revision of the GOLD guidelines made significant modifications to the combined assessment tool based on evidence that it failed to outperform basic spirometric classification. Furthermore, its complexity led the Workgroup to separate airflow limitation from the “ABCD” grades. Spirometry remains critical for the diagnosis of COPD and assessment of its severity; however, some treatment recommendations are based solely on symptoms and risk of exacerbations.
GOLD recognizes the possibility that there might be a marked disagreement between symptom severity predicted by the spirometric classification (GOLD 1, 2, 3, or 4) and the ABCD model. In such cases, they recommend more extensive testing to assess lung function (full pulmonary function tests), structural abnormalities (e.g., CT scans), and/or the presence of co-morbidities that might affect symptoms.
There is no evidence that this assessment strategy or any treatment based on it significantly improves meaningful COPD outcomes. In contrast, the ACP/ACCP/ATS/ERS guidelines do not propose a specific assessment strategy based on limitations in available evidence. Instead, they recommend treatment based on FEV1.4
GOLD recommends bronchodilators first line for the initial treatment of COPD
The GOLD guidelines recommend that all symptomatic patients with COPD should be prescribed short-acting bronchodilators for immediate rescue use. In addition, either short- or long-acting bronchodilators should be prescribed on a scheduled basis as they reduce breathlessness on a long-term basis. GOLD recommends basing the choice of therapy on the patient's risk of symptoms and future exacerbations as indicated by their ABCD group.1
Table 2. Preferred Initial Pharmacologic Therapy in Symptomatic Patients
| Category | Recommended treatment |
| GOLD Recommendations | |
| A Low Risk, Less Symptoms |
Preferred
|
| B Low Risk, More Symptoms |
Preferred:
|
| C High Risk, Less Symptoms |
Preferred
|
| D High Risk, More Symptoms |
Preferred
|
| ACP/ACCP/ATS/ERS Recommendations | |
| 60% ≤ FEV1 < 80% | Inhaled bronchodilators may be used |
| FEV1 < 60% | Inhaled bronchodilators should be used
|
| Adapted from: GOLD guidelines. URL: http://www.goldcopd.org/. Updated: 2020. Accessed: November 13, 2019. Qaseem A, et al. Ann Intern Med. 2011;155:179-191. Abbreviations: GOLD = Global Initiative for Chronic Obstructive Lung Disease, LABA = long-acting beta-agonist, LAMA = long-acting antimuscarinic, ICS = inhaled corticosteroid, CAT = COPD Assessment Test, ACP = American College of Physicians, ACCP = American College of Chest Physicians, ATS = American Thoracic Society, ERS = European Respiratory Society, FEV1 = forced expiratory volume in 1 second |
|
There is limited and conflicting evidence of the benefits of inhaled bronchodilators in symptomatic patients with less severe airflow obstruction. When the ACP reviewed studies in symptomatic participants with normal airflow or only mild-to-moderate COPD (e.g., FEV1 50-80% predicted), they found that chronic maintenance therapy provided little to no improvement in exacerbations, health status, hospitalizations, or deaths. However, the ACP/ACCP/ATS/ERS guidelines suggest that maintenance therapy may be used in these individuals as it might lead to an improvement in respiratory symptoms in some patients.2
GOLD recommends modifying COPD treatment based on a “review, assess, then adjust” process.
Follow up of patients on pharmacological treatment for COPD should be followed up systematically. The first step is to review the patient’s symptom burden and exacerbation risk. Before adjusting medications, assess the patient’s inhaler technique, adherence with the drug regimen, and consider whether non-pharmacological therapy has been optimized. Finally, adjust the medication regimen by escalating or de-escalation the intensity of therapy or by changing drugs or devices within the same class.2
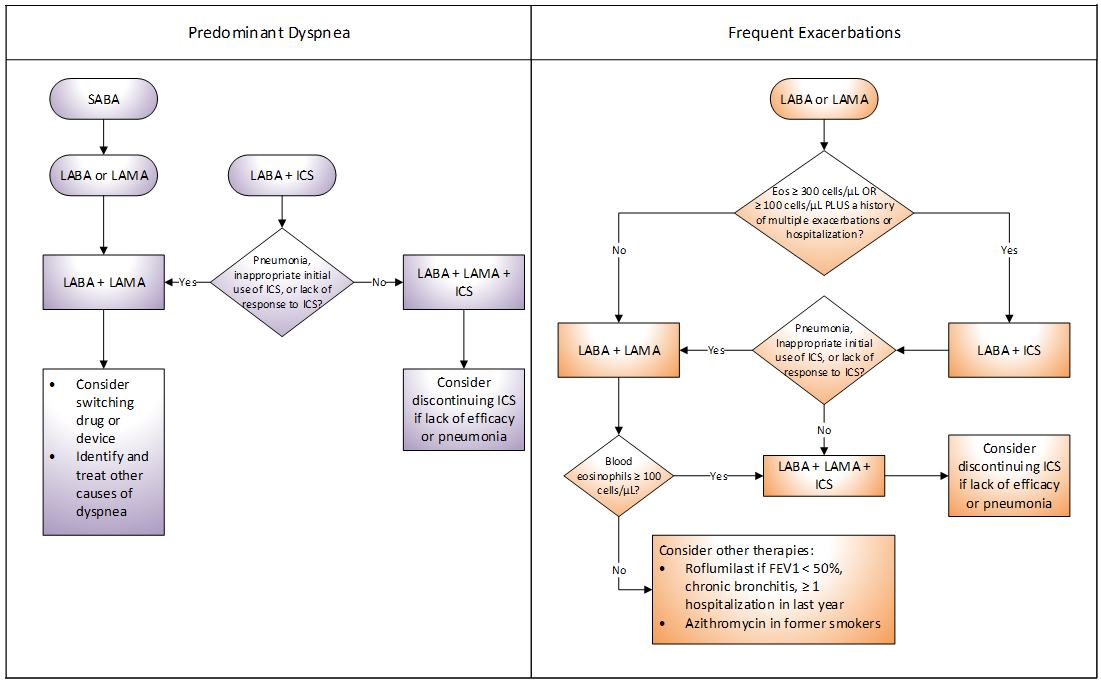
If patients respond favorably to therapy, continue it. Patients with an inadequate response to therapy will often present with one of two symptom profiles—predominant dyspnea/exercise limitation or frequent exacerbations. The GOLD guidelines recommend basing modifications of therapy on the presenting symptom profile. However, they acknowledge that there is an absence of evidence supporting this practice.1
Figure 3. GOLD Recommendations for Managing Patients with Inadequate Response to Therapy
Adapted from: GOLD guidelines. URL: http://www.goldcopd.org/. Updated: 2020. Accessed: November 13, 2019.
Add-on therapy with multiple medications is associated with high costs and adverse events, but has not been shown to consistently improve outcomes.
Combining inhaled therapies with different therapeutic actions, such as LABA/ICS or LABA/long-acting antimuscarinic (LAMA), typically results in greater improvements in lung function, symptoms, and exacerbations over monotherapy; however, not all improvements are clinically significant. The GOLD guidelines generally support the use of combination therapy in an effort to increase efficacy while ACP/ACCP/ATS/ERS concluded that combination therapy is not consistently superior to monotherapy. They suggest that the combination of a LABA plus ICS may be used in symptomatic patients with COPD and FEV1 < 60%, but the evidence is insufficient to support a strong recommendation for the broad use of combination therapy.2 The evidence continues to evolve, yet, to date, large systematic reviews and meta-analyses fail to consistently demonstrate clinically-significant benefits with combination therapy.7,8,9
The GOLD guidelines suggest that triple therapy (ICS plus LABA plus LAMA) may improve lung function and prevent exacerbations; however, data demonstrating clinically significant benefits are limited. Recent data suggest that, although triple therapy is associated with statistically significant reductions in exacerbations and improvements in quality of life compared to monotherapy or dual combination therapy, the magnitude of difference is generally small and of questionable clinical significance.10,11,12
Table 3. Selected Medications
| Generic (Brand) Name | Usual Dosea | Renal failure dosing? | Generic Avail?a | Discount Generic Program?b | Approx. Cost per Monthc |
Notes |
| Short-acting beta-agonists | ||||||
| Albuterol (Ventolin HFA) MDI | 1-2 puffs every 4-6 hours | No | Yes | No | $50 | Notable adverse events: Nervousness, tremor, chest pain, tachycardia, palpitation, headache, nausea, hyperglycemia |
| Short-acting anticholinergics | ||||||
| Ipratropium (Atrovent HFA) MDI | 2 puffs four times daily | No | No | No | $400 | Notable adverse events: Dizziness, headache, dry mouth, nausea, urinary tract infection, palpitations, tachycardia, constipation |
| Combination short-acting beta-agonist plus short-acting anticholinergic | ||||||
| Albuterol/ipratropium (Combivent Respimat) MDI |
1 puff four times daily | No | No | No | $400 | Notable adverse events: Headache, palpitations, tachycardia, tremor, constipation, nausea, vomiting |
| Long-acting beta-agonists | ||||||
| Salmeterol (Serevent Diskus) DPI | 1 puff every 12 hours | No | No | No | $400 | Notable adverse events: Headache, anxiety, muscle cramps, cough, rhinitis/sinusitis, hypertension |
| Indacaterol (Arcapta) DPI | 1 puff very 24 hours | No | No | No | $250 | Notable adverse events: Cough, nasopharyngitis, oropharyngeal pain, headache, nausea |
| Long-acting anticholinergics | ||||||
| Tiotropium (Spiriva Respimat) MDI | 2 puffs every 24 hours | No | No | No | $450 | Notable adverse events: Dry mouth, pharyngitis, chest pain, headache, urinary tract infection |
| Tiotropium (Spiriva HandiHaler) DPI | 1 puff every 24 hours | No | No | No | ||
| Aclidinium (Tudorza Pressair) DPI | 1 puff every 12 hours | No | No | No | $550 | Notable adverse events: Headache, diarrhea, cough, tachycardia |
| Umeclindinium (Incruse Ellipta) DPI | 1 puff every 24 hours | No | No | No | $350 | Notable adverse events: Tachycardia, atrial fibrillation, cough |
| Combination long-acting beta-agonist plus inhaled corticosteroid | ||||||
| Formoterol/budesonide (Symbicort) MDI | 2 puffs (160/4.5 mcg) every 12 hours | No | No | No | $350 | Notable adverse events: Bronchitis, nasopharyngitis, oral candidiasis, angina, palpitations, tachycardia, agitation, dizziness, pneumonia |
| Salmeterol/fluticasone (Advair Diskus) DPI | 1 puff (500/50 mcg) every 12 hours | No | No | No | $475 | Notable adverse events: Headache, cough, pharyngitis, palpitations, tachycardia, dizziness, nausea, vomiting, oral candidiasis, pneumonia |
| Salmeterol/fluticasone (Advair HFA) MDI | 2 puffs (460/42 mcg) every 12 hours | No | Yes | No | $150 | |
| Combination long-acting beta-agonist plus long-acting anticholinergic | ||||||
| Umeclindinium/vilanterol (Anoro Ellipta) DPI | 1 puff (62.5/25 mcg) every 24 hours | No | No | No | $400 | Notable adverse events: Cough, chest pain, arrhythmias |
| Glycopyrrolate/formoterol (Bevespi) MDI | 2 puffs (9/4.8 mcg) every 12 hours | No | No | No | $350 | Notable adverse events: Cough, urinary retention, urinary tract infection, EKG changes, hyperkalemia |
| Combination inhaled corticosteroid plus long-acting beta-agonist plus long-acting anticholinergic | ||||||
| Fluticasone/vilanterol/umeclidinium (Trelegy Ellipta) DPI | 1 puff once daily | No | No | No | $550 | Notable adverse events: Pharyngitis, dysphonia, candidiasis, sinusitis, headache, tachycardia, myalgia/arthralgia, nasopharyngitis |
| a Based on information from Facts & Comparisons. Some patients may require individualized dosing of certain medications. Refer to each drug’s prescribing information for detailed recommendations. b Substantially discounted generic medications (e.g., “$4 generics”) may be available through certain health plans or pharmacies. Individual lists are available from each retailer. c Approximate monthly cost estimated from wholesale acquisition cost (WAC). Actual pricing may vary. Abbreviations: MDI = metered-dose inhaler (aerosol), DPI = dry powder inhaler, EKG = electrocardiogram |
||||||
Palliative, end-of-life, and hospice care are important considerations in all patients with a life-limiting illness such as COPD.
The GOLD guidelines recommend palliative care in all patients with the goal of preventing and relieving suffering due to pulmonary and extra pulmonary symptoms (e.g., dyspnea, fatigue, depression, anxiety, insomnia) and improving quality of life. Palliative care is a broad term that includes everything from symptom management in an early-stage patient to management of patients close to death. For patients nearing the end of life, including those with progressively declining health, worsening symptoms, and increasingly frequent exacerbations, clinicians should initiate discussions on end-of-life care . Appropriate topics include resuscitation, advance directives, and their preferred terms of engagement with death. Hospice should be considered for patients who have severe disability or symptom burden.1
Patients with severe disease may gain solace in learning about palliative interventions, such as terminal sedation, which will protect them from experiencing discomfort at the end of life.4 The National Hospice and Palliative Care Organization (http://www.nhpco.org) can assist with the selection of patients with non-cancer diagnoses who might benefit from hospice services.
Self-management of exacerbations using a mutually agreed upon action plans may reduce the risk of hospitalization.
COPD exacerbations, defined as acute worsening of respiratory symptoms that require additional therapy, are associated with worsening health status, disease progression, hospitalization, and readmission. Mild exacerbations can be treated with short-acting bronchodilators alone, moderate exacerbations require antibiotic or systemic corticosteroid therapy, and severe exacerbations require emergency care or hospital admission (Table 4 ). Most exacerbations last for 7-10 days, but 20% of patients have not returned to their baseline after 2 months.1
The vast majority of exacerbations are managed in the outpatient settings, but patients should be assessed to determine whether they need acute care. Patients with acute respiratory failure, confusion or drowsiness, new physical signs or sudden worsening of dyspnea, failure of initial medical management, serious co-morbidities, or inability to manage the exacerbation at home should be considered for inpatient management.1
GOLD suggests that personalized self-management, including education, adherence to the care plan, symptom monitoring and management, and psychosocial coping, should be component of care for patients with COPD.1 For many patients, this also includes the early management of exacerbations at home. Based moderate-to-high quality evidence from 22 studies (3854 participants), the Cochrane Collaboration reports that multi-component self-management interventions that include an action plan for exacerbations may prevent one respiratory hospitalization for every 12 patients treated and marginally improve quality of life (-2.7 points on the St. Georges Respiratory Questionnaire [SGRQ]; minimum clinically important difference 4 points). There was also an increase in respiratory-related mortality compared to usual care (9% vs. 5%); however, the evidence was of very low quality.13 Even without a self-management intervention, Cochrane reports that action plans with a brief education component may prevent one hospitalization for every 19 people treated one and one emergency department visit for every 12 treated for one year along with a comparably small improvement in quality of life (-2.8 points on the SGRQ) compared to usual care (7 studies, 976 participants).14
Pulmonary rehabilitation can reduce the risk of re-hospitalization and death among patients admitted with a COPD exacerbation.
Patients hospitalized for a COPD exacerbation should be followed up promptly within one month of discharge and again at 3-4 months to ensure that they are clinically stable. Follow up should include the following:1
- Review of the exacerbation
- Documentation of symptoms
- Review of the medication regimen and assessment of inhaler technique
- Assessment of the need for long-term oxygen therapy
- Evaluation of the patient’s burden of comorbidities and their ability to perform activities of daily living and cope in their current environment
Evidence suggests that hospitalization for a COPD exacerbation should be considered a trigger for assessment for pulmonary rehabilitation. The GOLD guidelines suggest that, if implemented within 4 weeks of discharge, pulmonary rehabilitation can reduce the risk or readmission and death [LOE: B].1
Table 4. Management of Acute COPD Exacerbations
| Treatment category | Interventions | Comments |
| Standard management for all exacerbations | ||
| Inhaled bronchodilators | Short-acting beta2-agonists +/- Short-acting anticholinergics |
Recommended for initial therapy for COPD exacerbations [LOE: C] |
| Corticosteroids | Prednisone 40 mg daily x 5 days | Improves FEV1 and hypoxia, shortens recovery, reduces the risk of treatment failure, and reduces hospital length of stay [LOE: A] |
| Antibiotics | Coverage for common bacterial respiratory pathogens (≤ 5-7 days) | Controversial due to limitations in the evidence. In patients with increased dyspnea, sputum volume, and sputum purulence , antibiotics may shorten recovery, reduce the risk of treatment failure, and reduce hospital length of stay [LOE: B] |
| Education | COPD exacerbation action plan | Shown to reduce in-hospital care utilization when supplemented with ongoing support |
| Hospital management | ||
| Oxygen support | Supplemental oxygen | Administer via nasal cannula or face mask to achieve an O2 saturation 88-92% |
| Ventilator support | Mechanical ventilation | Non-invasive ventilation is recommended as first-line mechanical ventilation for all patients with acute respiratory failure who do not have contraindications |
| Adapted from: GOLD guidelines. URL: http://www.goldcopd.org/. Updated: 2020. Accessed: November 13, 2019. Abbreviations: COPD = Chronic Obstructive Pulmonary Disease, FEV1 = forced expiratory volume in 1 second |
||